Sputtering Spray Method – Background Entering the 21st century, with the improvement of environmental protection requirements, the rise in energy costs and the upgrading of demands in segmented scenarios, sodium hypochlorite production technology has begun to optimize towards the direction of "high efficiency, energy saving, low by-products and customization". |  |
Sputtering Spray Method for Sodium Hypochlorite Preparation First, catalytic materials such as ruthenium (Ru) and iridium (Ir) are deposited as nano-coatings on a titanium sheet substrate via sputtering to produce electrodes with high activity and durability. Then, this electrode is used as the anode and placed in an electrolyzer. After energization, Cl⁻ in the brine is catalyzed by the electrode to generate Cl₂, which then reacts with OH⁻ in the solution to produce sodium hypochlorite. The entire process achieves low energy consumption and increased output. | 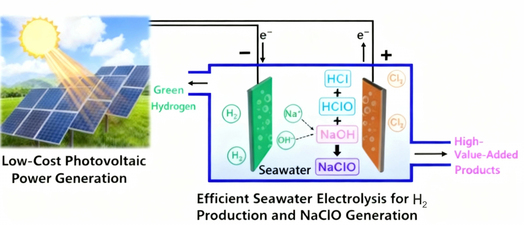
Schematic diagram of sputtering spray reaction |
Significance of the Sputtering Spray Reaction
The Ru-based ternary coated titanium anode prepared by the sputtering spray method has a coating with uniform thickness. Its microstructure exhibits numerous cracks, which is conducive to accelerating mass transfer and promoting chlorine gas evolution. At 200 mA/cm², the chlorine evolution potential, oxygen evolution potential, and oxygen-chlorine potential difference of this anode are 1.098 V, 1.397 V, and 0.299 V respectively. It features a low degree of oxygen evolution side reactions and high efficiency in electrolytic sodium hypochlorite production.

 |
Advantages Reduce electrolysis energy consumption and improve hydrogen production efficiency. Extend electrode service life and reduce replacement costs. Improve sodium hypochlorite purity and reduce by-products. Adapt to flexible scenarios and lower application thresholds. |