Comprehensive Analysis of Hydrogen Production Technologies in the Green Hydrogen Industry Chain
Hydrogen is both an industrial gas and an energy carrier, with applications in more than 20 fields. China, which produces and consumes over 30 million tons of hydrogen annually, is a major hydrogen producer and consumer. Thanks to its advantages such as storability, renewability, complete zero-carbon production potential, and high energy density, hydrogen energy has been playing an increasingly important role in the energy structures of countries around the world. However, the vast majority of hydrogen production currently relies on fossil fuels and by-product hydrogen, resulting in additional carbon dioxide emissions. Obviously, this fails to meet the requirements of the dual carbon goals and is inconsistent with the vision of green development for humanity.
Mainstream Water Electrolysis Hydrogen Production Technologies
Currently, there are four mainstream technological routes for water electrolysis hydrogen production: Alkaline Water Electrolysis (ALK), Solid Oxide Electrolysis Cell (SOEC), Anion Exchange Membrane (AEM) electrolysis, and Proton Exchange Membrane (PEM) electrolysis, which is on the verge of commercialization. Faced with carbon emission reduction pressures, water electrolysis hydrogen production is perhaps the closest to zero-carbon emission technology among industrial hydrogen production methods available today. The iterative application of water electrolysis hydrogen production technology will directly reduce carbon emissions in both traditional and emerging fields such as chemical engineering, petrochemicals, transportation, and hydrogen metallurgy.
Alkaline Water Electrolysis (ALK)
Hydrogen energy is a relatively uncommon clean energy source. During its use, it produces no additional pollution or carbon dioxide emissions. Moreover, hydrogen has an energy density as high as 140 MJ/kg, which is 4.5 times that of coal and 3 times that of oil. As an energy source with both cleanliness and high efficiency, hydrogen energy is currently regarded as one of the potential ultimate energy sources for achieving a zero-carbon society. Since the issuance of the "Medium and Long-Term Plan for the Development of the Hydrogen Energy Industry (2021-2035)" in 2022, the strategic industrial position of hydrogen energy in China has been basically established. Up to now, more than 20 provinces across the country have issued over 200 hydrogen energy plans and guiding opinions, with more than 30 large-scale industrial enterprises in the entire industrial chain. The hydrogen energy industry in China has gradually formed several powerful cluster forces in three major regions: the Yangtze River Delta, the Guangdong-Hong Kong-Macao Greater Bay Area, and the Bohai Rim.
Principle: Direct current is passed through a high-concentration potassium hydroxide solution, causing water molecules to undergo electrochemical reactions at the electrodes. Water molecules decompose into hydrogen ions (H⁺) and hydroxide ions (OH⁻) at the cathode. Hydrogen ions combine with electrons from the cathode to form hydrogen gas, while hydroxide ions move to the anode to generate oxygen and water.
Disadvantages: High energy consumption – alkaline water electrolysis requires a large amount of electrical energy and has relatively low electrolysis efficiency; under alkaline conditions, the electrodes in the electrolyzer are prone to corrosion, leading to shortened equipment lifespan and increased maintenance costs; poor membrane stability under alkaline conditions – the alkaline environment places high demands on membrane stability, and membrane damage will reduce electrolysis efficiency; relatively low hydrogen purity – the hydrogen produced by alkaline water electrolysis has relatively low purity, requiring additional treatment steps to obtain higher-purity hydrogen. Due to the maturity of the product, low energy consumption and large standard cubic meters have become the mainstream research directions. According to High-Tech Hydrogen Power reports, the cost of a single-tank hydrogen production equipment with large standard cubic meters is lower than purchasing multiple sets of equipment with the same output. In 2022, 1000 Nm³/h alkaline electrolyzers have become mature, with a total domestic single-tank capacity of 15,700 Nm³/h; the current average power consumption in the industry is 4.5-4.6 kWh/Nm³, and reducing the DC power consumption of electrolyzers is the focus of electrolyzer technology upgrading.
Solid Oxide Electrolysis Cell (SOEC)
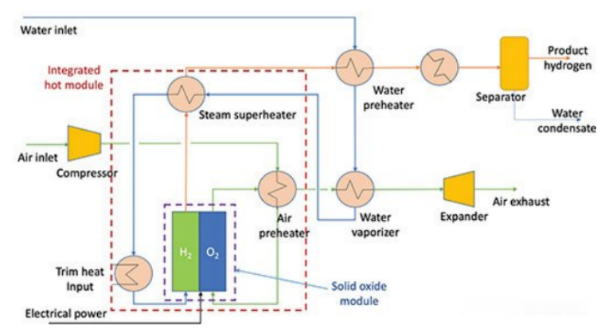
The principle of SOEC is to electrolyze water vapor to produce hydrogen under high-temperature conditions, with the overall reaction being 2H₂O → 2H₂ + O₂. The SOEC electrolyzer consists of a hydrogen electrode layer, an electrolyte layer, and an oxygen electrode layer. The hydrogen electrode is a porous ceramic structure (e.g., Ni-YSC cermet) responsible for conducting electrons and transporting water vapor and generated hydrogen; the electrolyte is a dense perovskite ceramic (e.g., YSZ) that can conduct oxygen ions; the oxygen electrode is a porous ceramic structure (e.g., LSM) responsible for conducting electrons and transporting air and generated oxygen.
Solid Oxide Hydrogen Production Process
Advantages: Compared with other water electrolysis methods, proton exchange membrane (PEM) water electrolysis hydrogen production technology boasts higher operating current density and electrolysis efficiency. It features a pollution-free reaction process, a lightweight and compact structure (with a smaller volume under the same power output), and the produced hydrogen can reach a purity of 99.999%. Thus, it is regarded as the most promising water electrolysis technology.
Disadvantages: Due to the high price of raw materials such as noble metals and the high difficulty of the production process, proton exchange membrane products are costly. Additionally, the proton exchange membrane used in water electrolysis hydrogen production is relatively thick, making its cost several times higher than that of proton exchange membranes applied in other fields. Undoubtedly, the PEM water electrolysis hydrogen production market, which is on the dawn of commercialization, has broad prospects. Based on data such as hydrogen demand and water electrolysis hydrogen production penetration rate from authoritative institutions, combined with the growth expectation of PEM's proportion in water electrolysis hydrogen production driven by domestic substitution and technological upgrading.
As a clean energy production method, water electrolysis green hydrogen holds enormous potential. Renewable energy power generation has opened up the ceiling for hydrogen energy development. In the future, with continuous technological progress and further cost reduction, the demand for green hydrogen is expected to usher in "high-slope" growth, making significant contributions to achieving the clean energy transition and carbon neutrality goals!
Source: Aibang Hydrogen Energy
Advantages: The biggest advantage of SOEC technology is its low power consumption, making it suitable for concentrated solar power systems that produce high-temperature and high-pressure steam. It has a wide range of applications: in addition to water electrolysis for hydrogen production, SOEC electrolyzers can also synthesize fuels with carbon dioxide and be used in fuel cells through reversible reactions. Furthermore, SOEC offers additional advantages such as good stability and high hydrogen production purity.
Disadvantages: The high-temperature and high-humidity operating environment limits the selection of electrolyzer materials that require high stability, durability, and anti-attenuation properties, which also restricts the application scenarios and large-scale promotion of SOEC hydrogen production technology. Durability is a key challenge for SOEC currently; thermochemical cycles, especially during system shutdown and startup, accelerate aging and reduce service life.
Current Status: The international SOEC market has entered the initial stage of industrialization, with representative enterprises including Sunfire (Germany), Bloom Energy, Fuelcell Energy, Cummins (USA), Topsoe (Denmark), etc.
Anion Exchange Membrane (AEM) Electrolysis
AEM utilizes the positive electric field formed by positively charged groups to facilitate ion transfer in the electrolyte while blocking the passage of cations. It also involves two half-reactions: the Oxygen Evolution Reaction (OER) and the Hydrogen Evolution Reaction (HER).
Structure: Composed of an anion exchange membrane and two transition metal catalytic electrodes, with pure water or a low-concentration alkaline solution as the electrolyte. The anion exchange membrane, a critical component, functions to conduct OH⁻ from the cathode to the anode while preventing the direct transfer of gases and electrons between the electrodes. After loading the catalyst onto the electrodes, the system is hermetically assembled with components such as bipolar plates, gas diffusion layers, and gaskets.
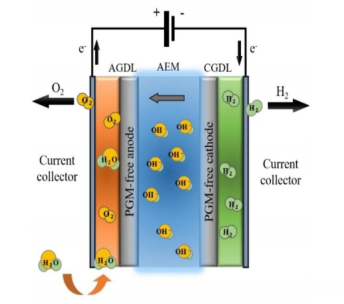
Schematic Diagram of the Operating Process of AEM Water Electrolysis for Hydrogen Production
Advantages: AEM integrates the merits of ALK and PEM. Non-noble metal catalysts can be employed in alkaline media; besides excellent air tightness and low electrical resistivity, the anion exchange membrane eliminates the need for expensive perfluorosulfonic acid membranes. Furthermore, AEM can use weak alkali or pure water as the electrolyte, mitigating equipment corrosion and product gas contamination issues caused by strong alkaline solutions.
Disadvantages: The anion exchange membrane exhibits poor thermal and chemical stability, along with limited anion conductivity, failing to meet the requirements of long-term operation. Secondly, large-scale production and cost reduction of AEM water electrolysis hydrogen production technology remain key challenges to be addressed.
Physical Image of EVE Energy's AEM Membrane Products

Proton Exchange Membrane (PEM) Electrolysis
Principle: In a PEM water electrolyzer, water is split at the anode into oxygen (O₂), electrons (e⁻), and protons (H⁺), with oxygen released from the anode. Electrons flow to the cathode through an external circuit, while protons migrate to the cathode via the proton exchange membrane. On the cathode side, two protons combine with electrons to produce hydrogen (H₂).
Structure: PEM electrolyzers adopt a proton exchange membrane to replace the diaphragm and liquid electrolyte in alkaline electrolyzers, fulfilling the dual functions of ion conduction and gas separation. Regarding the cost composition of PEM electrolyzers: 45% is allocated to the electrolytic stack, and 55% to system auxiliary equipment; among the electrolytic stack cost, 24% is accounted for by the membrane electrode.
Schematic Diagram of PEM Water Electrolysis Hydrogen Production Structure
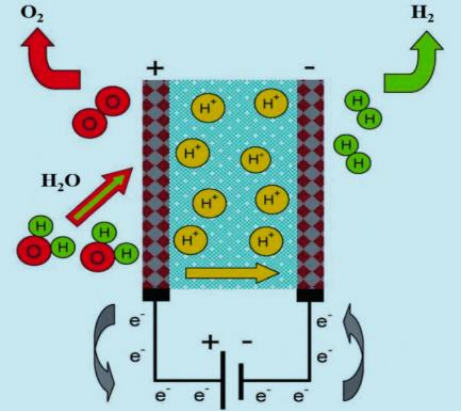
Advantages: Compared with other water electrolysis methods, proton exchange membrane (PEM) water electrolysis hydrogen production technology boasts higher operating current density and electrolysis efficiency. It features a pollution-free reaction process, a lightweight and compact structure (with a smaller volume under the same power output), and the produced hydrogen can reach a purity of 99.999%. Thus, it is regarded as the most promising water electrolysis technology.
Disadvantages: Due to the high price of raw materials such as noble metals and the high difficulty of the production process, proton exchange membrane products are costly. Additionally, the proton exchange membrane used in water electrolysis hydrogen production is relatively thick, making its cost several times higher than that of proton exchange membranes applied in other fields. Undoubtedly, the PEM water electrolysis hydrogen production market, which is on the dawn of commercialization, has broad prospects. Based on data such as hydrogen demand and water electrolysis hydrogen production penetration rate from authoritative institutions, combined with the growth expectation of PEM's proportion in water electrolysis hydrogen production driven by domestic substitution and technological upgrading.
As a clean energy production method, water electrolysis green hydrogen holds enormous potential. Renewable energy power generation has opened up the ceiling for hydrogen energy development. In the future, with continuous technological progress and further cost reduction, the demand for green hydrogen is expected to usher in "high-slope" growth, making significant contributions to achieving the clean energy transition and carbon neutrality goals!
Article Source: Official WeChat Account "New Energy Technology and Equipment"
Disclaimer: The information reposted on this website is sourced from cooperative media, institutions, or other websites. The purpose of publishing this article is to convey more information, and it does not imply endorsement of its views or confirmation of the authenticity of its content. All information on this website is for reference only and shall not be used as the basis for transactions or services. If any content on this website is infringing or has other issues, please notify us promptly, and we will modify or delete it in a timely manner. Anyone who accesses this website in any way or directly/indirectly uses the materials on this website is deemed to voluntarily accept the constraints of this website's statement. The copyright of this article belongs to the official WeChat Account "New Energy Technology and Equipment".