Hydrogen Production
When discussing hydrogen energy, a common misconception is that it is an energy source that can be directly extracted, much like underground petroleum or coal. In fact, hydrogen is the most abundant element in the universe, yet it rarely exists in its pure form. It is more accurately described as an energy carrier that must be "produced" from hydrogen-containing substances such as water and fossil fuels through various technologies. Therefore, "hydrogen production" is undeniably the source and starting point of the entire hydrogen energy industry chain.
Hydrogen energy cannot be directly obtained through exploration and extraction like petroleum and coal, but must be produced via hydrogen generation technologies. Hydrogen production is the first link in the hydrogen energy industry chain. At present, the three main large-scale applicable hydrogen production routes are as follows: first, fossil energy-based hydrogen production dominated by coal and natural gas conversion; second, industrial by-product-based hydrogen production including coke oven gas, chlor-alkali tail gas, and propane dehydrogenation; third, water electrolysis-based hydrogen production. Other hydrogen production technologies such as biomass-derived hydrogen production and solar-driven photolytic water splitting are yet to be commercialized on a large scale.
Fossil Energy-Based Hydrogen Production
Fossil energy-based hydrogen production technologies such as coal-to-hydrogen and natural gas-to-hydrogen are the primary methods for industrial hydrogen production. Coal-to-hydrogen refers to a process where coal reacts with steam or oxygen at high temperature and normal pressure, or high temperature and high pressure, to generate hydrogen and carbon monoxide first; then, carbon monoxide undergoes the water-gas shift reaction to produce hydrogen and carbon dioxide. The hydrogen production process includes coal gasification, carbon monoxide water-gas shift, acid gas removal, hydrogen purification and waste treatment. After purification, hydrogen with a purity of 99.9% can be obtained.Coal-to-hydrogen technology boasts advantages including abundant raw materials, low costs and mature technology, accounting for the largest share of hydrogen production in China. However, this method is accompanied by substantial carbon dioxide emissions.
Natural gas is a gaseous fossil fuel composed primarily of methane. It is mainly found in oilfields and natural gas fields, with small amounts also present in coal seams. Currently, there are four main technical routes for natural gas-based hydrogen production: steam methane reforming, partial oxidation reforming of natural gas, autothermal reforming of natural gas, and high-temperature pyrolysis of natural gas. Natural gas-based hydrogen production offers considerable economic benefits and has evolved into one of the mainstream technologies for industrial hydrogen production.
Industrial By-product-based Hydrogen Production Industrial by-product-based hydrogen production refers to the utilization of hydrogen resources generated as by-products during industrial processes such as coke oven gas processing, chlor-alkali chemical production, ammonia synthesis, methanol synthesis, ethylene cracking, and propane dehydrogenation. This method boasts advantages including wide-ranging hydrogen sources, low investment, mature technology, and low costs. By-product hydrogen can be purified into high-purity hydrogen through processes like pressure swing adsorption (PSA) and deep desulfurization, and most of it has already been applied in downstream sectors at present.
Coke oven gas has great potential for hydrogen supply. It features large-scale production and relatively low production costs. However, coke oven gas has a low hydrogen content and contains considerable harmful impurities such as carbon monoxide and sulfides, leading to high purification and desulfurization costs. Thus, it is not very suitable for fuel cells that require high-purity hydrogen. The by-product hydrogen from chlor-alkali chemical production has high purity and few harmful impurities, making it suitable for fuel cell applications. Nevertheless, chlor-alkali chemical enterprises are relatively scattered, with a small hydrogen supply volume per enterprise. Traditional chemical industries such as ammonia synthesis and methanol synthesis produce a large amount of tail gas that is directly vented into the air; the tail gas contains 18%–55% hydrogen. The by-product hydrogen from propane dehydrogenation has a high hydrogen content and few carbon monoxide impurities, but it still requires deep desulfurization treatment.
Water Electrolysis for Hydrogen Production
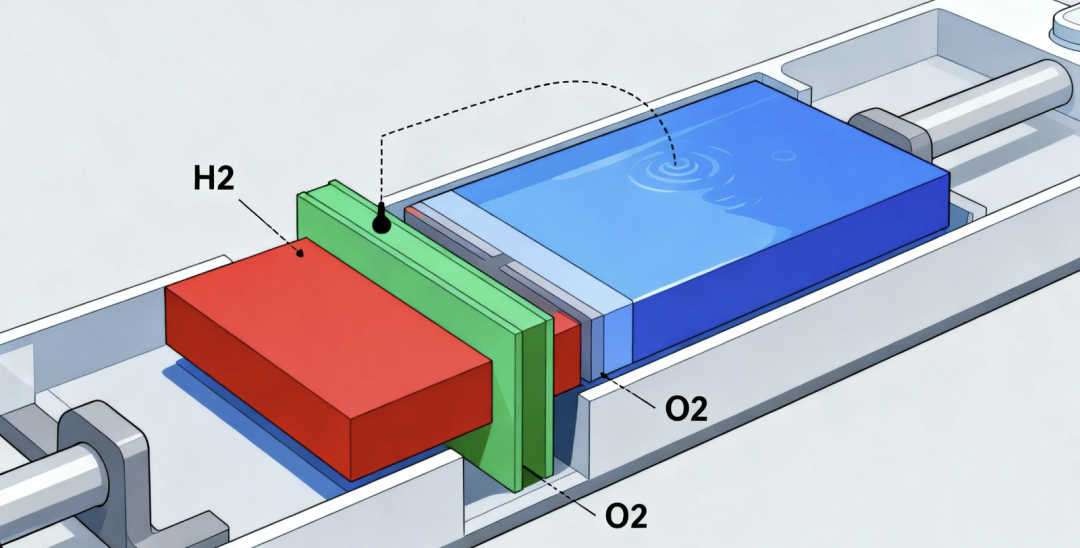
The basic principle of water electrolysis for hydrogen production is to use water as the raw material and apply an external voltage to form a complete electrical circuit. The input of electrical energy breaks the internal balance of water molecules, causing them to decompose. Hydrogen and oxygen atoms then recombine, eventually leading to the evolution of hydrogen and oxygen gases. A water electrolysis hydrogen production system mainly consists of modules such as the electrolyzer, power conversion, water circulation, gas separation, and gas purification. As the core component of the system, the electrolyzer is the primary site for the electricity-to-hydrogen conversion reaction, and it is composed of three parts: electrolyte, diaphragm, and electrodes.
Water electrolysis technology features simple processes, easy operation, environmental friendliness, low carbon emissions, and high hydrogen purity. Compared with other hydrogen production methods, it uses water as the raw material and can achieve the goal of zero-pollution hydrogen production to the greatest extent. Currently, there are four main technical routes for water electrolysis hydrogen production: alkaline water electrolysis (ALK), proton exchange membrane water electrolysis (PEM), solid oxide electrolysis (SOE), and anion exchange membrane water electrolysis (AEM).
Hydrogen Energy Development: From Low-Carbon to Zero-Carbon
Looking at the development landscape of hydrogen energy, its evolution path—from the current mainstream fossil energy-based hydrogen production to the ideal renewable energy-powered water electrolysis—clearly points to a single core: low-carbon and zero-carbon. As a vital bridge connecting fossil energy and renewable energy, hydrogen energy has thus defined its ultimate role in the global energy revolution.
In the future, as the cost barriers to hydrogen production are gradually overcome, hydrogen energy will surely expand from being an industrial raw material to a clean energy carrier that supports grid balancing, empowers heavy-duty transportation, and achieves deep decarbonization in industries. It is not only a key piece in realizing the "dual carbon" goals but also a core driving force for building a sustainable energy system of the future.
Source: ZeroCarbonCity
Disclaimer:The information published on this website is reproduced from cooperative media, institutions or other third-party platforms. The publication of this article is for the purpose of disseminating more information only, and does not imply endorsement of its views or verification of the authenticity of its content. All information on this website is provided for reference only, and shall not be used as the basis for any transactions or services.Should any content on this website be deemed infringing or otherwise problematic, please notify us promptly and we will modify or remove the relevant content in a timely manner. Any person who accesses this website in any form or uses the materials on this website directly or indirectly shall be deemed to have voluntarily accepted the terms and constraints of this disclaimer. All copyrights belong to the original authors.